 Critical aspects (CAs) are identified through system risk assessments. 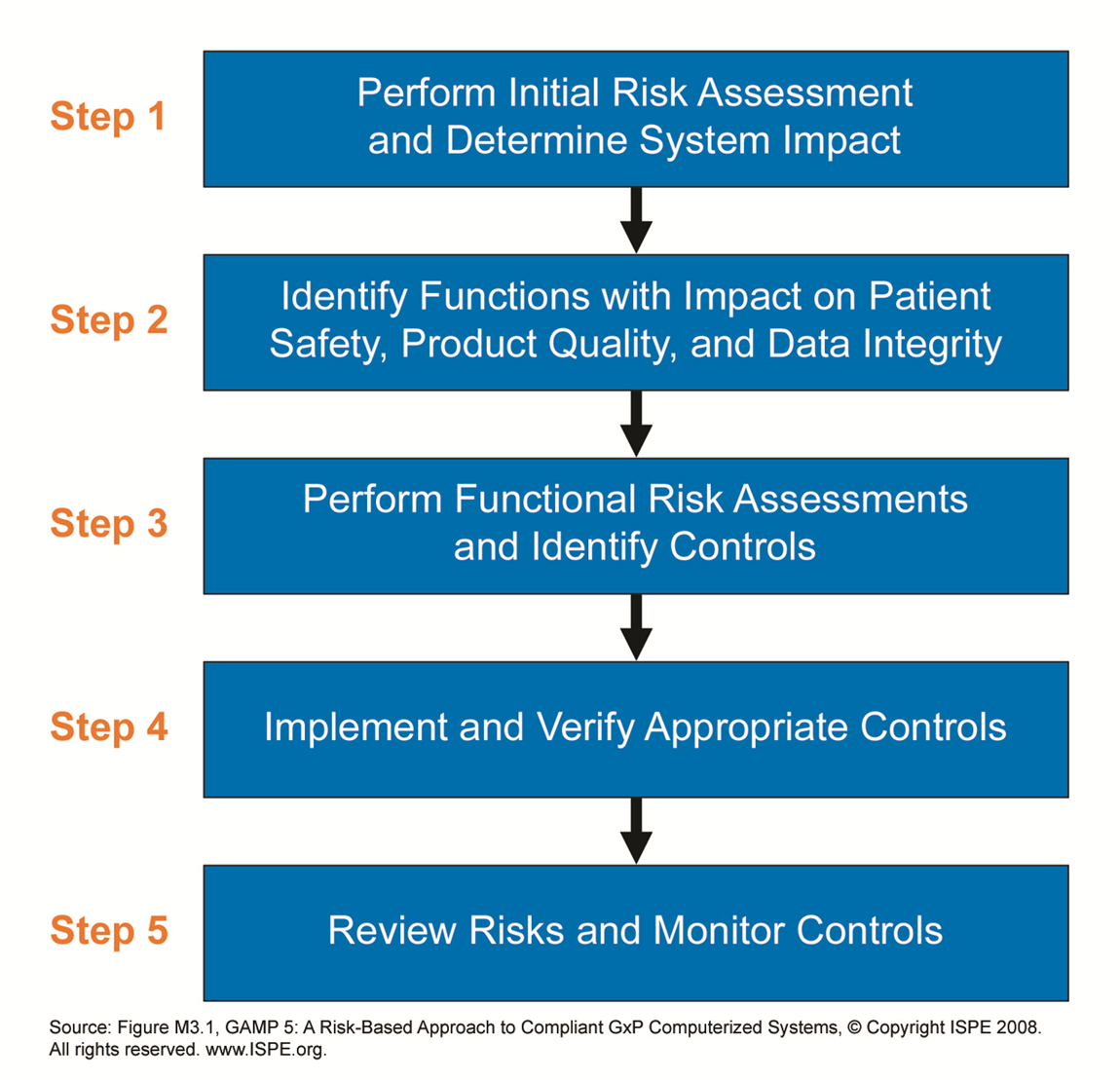
(Ch3)Ĭritical Design Elements (CDEs) are usually identified based on technical understanding of the products critical quality attributes, process critical process parameters, and equipment/automaton design. User Requirements Specifications: Is it necessary to define Critical Design Elements and critical process parameters during the preparation of user requirement specifications?Ĭritical quality attributes and critical process parameters are key inputs into user requirements specifications, and the quality risk management commissioning and qualification process, and should be identified prior to user requirements specifications generation. Laboratory support equipment, such as controlled temperature storage units, and critical utilities serving laboratories, such as USP/WFI water and gases are covered in Guide Scope. Laboratory instruments are not in the scope of the Guide. Can the Guide approach be applied to lab instruments including any associated automation? GAMP® describes a science and risk based approach, and the GAMP® organization are always looking for ways to optimize the approach. IT and IS are out of the scope of the Guide and fall under GAMP®. What is the impact of the Guide on IT and IS systems, (i.e., ERP, SAP, etc.)?  
These aligned user requirements are verified using an integrated testing strategy. For automation/Process Control Systems attached to systems and equipment the user requirements specifications for each must align when addressing critical process parameter control, alarm management, and data management. GAMP® describes a science risk-based approach for hardware and software development. All other computerized systems fall under GAMP®. The scope of the BG5 revision is equipment and automated systems. What is the alignment between the new Guide and GAMP® 5?
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
